Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (3): 396-403.doi: 10.3969/j.issn.2095-4344.0599
Previous Articles Next Articles
Protective effects of methane saturated saline on endotoxin-induced acute lung injury in rats
Meng Qingxi1, Wang Weiheng2, Sun Aijun3, Xi Yanhai1, Ye Xiaojian1
- (1Center for Minimally Invasive Spine Surgery, Shanghai Changzheng Hospital Affiliated to the Second Military Medical University, Shanghai 200433, China; 2Shanghai Changzheng Hospital Affiliated to the Second Military Medical University, Shanghai 200433, China; 3Department of Anatomy and Histoembryology, Second Military Medical University, Shanghai 200433, China)
-
Received:2018-07-28Online:2019-01-28Published:2019-01-28 -
Contact:Ye Xiaojian, MD, Professor, Chief physician, Center for Minimally Invasive Spine Surgery, Shanghai Changzheng Hospital Affiliated to the Second Military Medical University, Shanghai 200433, China -
About author:Meng Qingxi, Doctoral candidate, Associate chief physician, Center for Minimally Invasive Spine Surgery, Shanghai Changzheng Hospital Affiliated to the Second Military Medical University, Shanghai 200433, China
CLC Number:
Cite this article
Meng Qingxi, Wang Weiheng, Sun Aijun, Xi Yanhai, Ye Xiaojian. Protective effects of methane saturated saline on endotoxin-induced acute lung injury in rats[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(3): 396-403.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
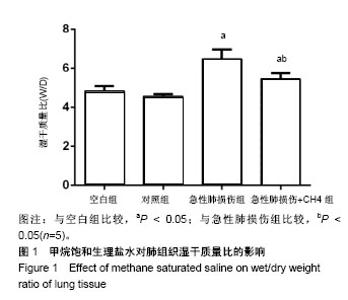
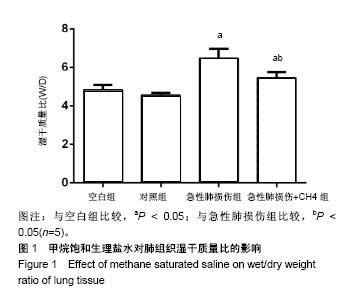
2.1 实验动物数量分析 实验选用SD大鼠40只,分为4组,实验过程无脱失,全部进入结果分析。 2.2 甲烷饱和生理盐水显著降低急性肺损伤大鼠肺组织W/D 内毒素及甲烷饱和生理盐水处理大鼠24 h后,取右肺检测肺组织的W/D,结果见图1,空白组与对照组相比未见明显差异(P > 0.05)。急性肺损伤组和急性肺损伤+CH4组肺组织的W/D明显高于空白组(P < 0.05),而急性肺损 伤+CH4组的肺组织W/D比明显低于急性肺损伤组(P < 0.05),且低于空白组(P < 0.05)。表明内毒素可以显著升高肺组织的W/D,而甲烷饱和生理盐水可以显著降低内毒素处理后急性肺损伤大鼠的肺W/D,且对正常肺组织的W/D比无显著影响。"
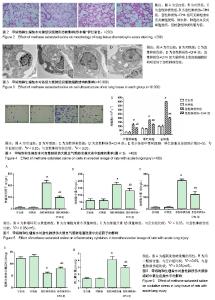
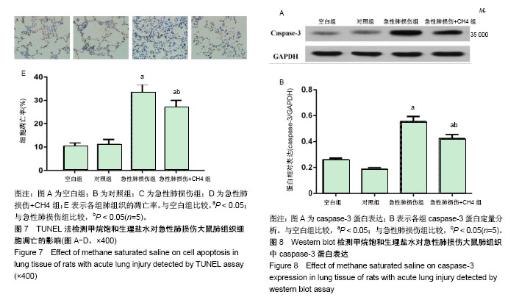
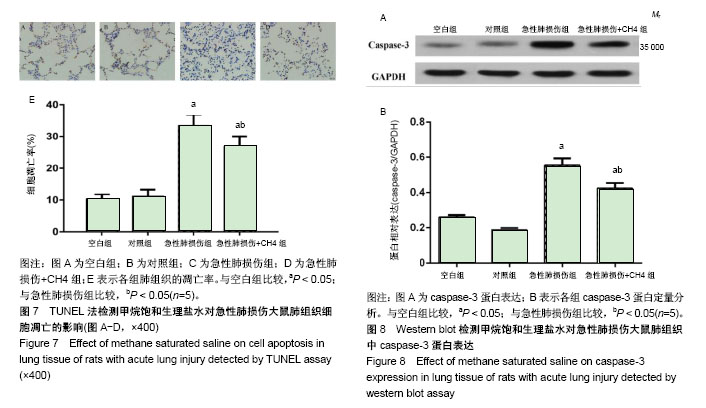
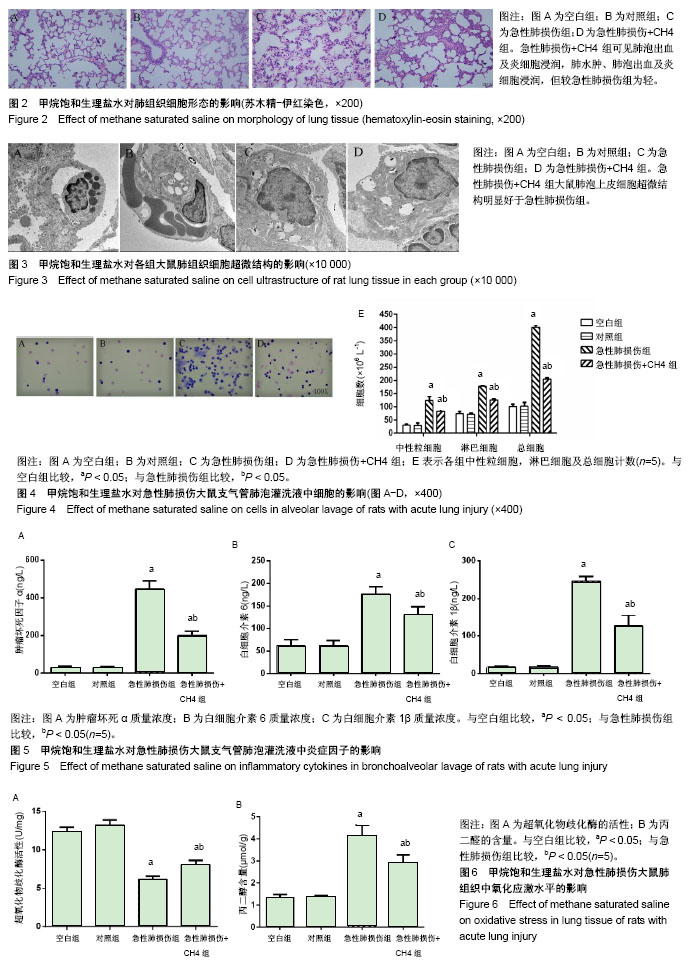
2.3 肺组织的苏木精-伊红染色和电子显微镜检测 内毒素处理1 h后大鼠开始出现活动度减少,精神差,痢疾等反应。急性肺损伤+CH4组与急性肺损伤组相比,上述症状较为轻微,大鼠活动度及精神状态尚可。组织学检查大体观察可见空白组和对照组大鼠肺组织表面光滑,呈粉红色,无液体渗出及出血点。光学显微镜下可见肺组织结构清晰,肺泡壁薄,肺泡内未见液体渗出,见图2A,B。电镜下观察肺泡的超微结构可见肺泡上皮细胞具有典型的线粒体及内质网结构,见图3A,B。内毒素处理组大鼠肺组织大体观察可见整个肺部充血水肿,表面粗糙,间有点状斑片状坏死。光学显微镜下见肺间质水肿,弥漫性出血,广泛炎性细胞浸润,肺泡壁增厚,内有大量液体渗出,见图2C。电镜下可见1型肺泡细胞出现典型的空泡形成,去颗粒,线粒体肿胀,核周池肿胀内质网变粗糙,2型肺泡上皮细胞可见典型的板层小体空泡和管状髓鞘脱落,见图3C。急性肺损伤+CH4组大鼠肺组织大体可见中度肺水肿,表面点状坏死灶,光学显微镜下可见肺泡出血及炎细胞浸润,肺水肿、肺泡出血及炎细胞浸润但较急性肺损伤组为轻,见图2D。电镜下肺泡上皮细胞超微结构明显好于急性肺损伤组,见图3D。 2.4 甲烷饱和生理盐水对急性肺损伤大鼠支气管肺泡灌洗液细胞的影响 见图4A所示,生理状况下大鼠支气管肺泡灌洗液中主要的细胞为中性粒细胞、巨噬细胞及淋巴细胞。甲烷饱和生理盐水处理对正常支气管肺泡灌洗液中细胞数量和种类影响不大(P > 0.05),见图4B。内毒素处理后,支气管肺泡灌洗液中细胞总量明显增加(P < 0.05),见图4C,其中以中心性粒细胞和巨噬细胞增多为主,而甲烷饱和生理盐水处理可以明显降低内毒素导致的支气管肺泡灌洗液中细胞总量及中性粒细胞和巨噬细胞的数量(P < 0.05),见图4D,E。 2.5 甲烷饱和生理盐水对急性肺损伤大鼠支气管肺泡灌洗液中炎症因子的影响 见图5所示,对照组和空白组相比炎症因子质量浓度差异无显著性意义(P > 0.1),表明富甲烷生理盐水对正常大鼠肺部的炎症因子无显著影响;急性肺损伤组和急性肺损伤+CH4组的支气管肺泡灌洗液中肿瘤坏死因子α、白细胞介素6和白细胞介素1β的质量浓度显著上升(P < 0.05),而经甲烷饱和生理盐水治疗后的急性肺损伤+CH4组与急性肺损伤组相比各炎症因子的质量浓度有显著降低(P < 0.05),但仍明显高于对照组和空白组(P < 0.05)。说明饱和甲烷生理盐水可以通过抑制肺部炎症因子的产生和分泌,降低由于内毒素导致的急性肺损伤。 2.6 甲烷饱和生理盐水对急性肺损伤大鼠肺组织氧化应激的影响 各组处理24 h后,肺组织中丙二醛和超氧化物歧化酶的含量测定结果见图6所示,空白组与对照组相比,丙二醛和超氧化物歧化酶的含量差异无显著性意义(P > 0.1)。急性肺损伤和急性肺损伤+CH4组与空白组相比肺组织中丙二醛含量显著升高,超氧化物歧化酶的含量显著降低(P < 0.05);急性肺损伤+CH4组与急性肺损伤组相比,丙二醛的含量显著降低,而超氧化物歧化酶含量显著升高(P < 0.05),但仍然未达到空白组与对照组水平(P < 0.05)。表明甲烷对正常大鼠肺组织中的丙二醛和超氧化物歧化酶的含量无显著影响,而急性肺损伤造模后,肺组织中丙二醛的含量显著增高,而超氧化物歧化酶含量显著降低。甲烷治疗急性肺损伤可以显著的降低肺组织中丙二醛含量,提高超氧化物歧化酶含量。 2.7 甲烷饱和生理盐水对急性肺损伤大鼠肺组织凋亡的影响 如图7所示,TUNEL染色时,正常细胞染色后细胞核蓝绿色,而凋亡细胞的细胞核被染成黄褐色。肺组织TUNEL染色结果显示:各组均可见棕黄色、棕褐色的凋亡细胞核,但Blank与对照组棕黄色细胞核出现率很低,细胞凋亡较少,且两组差异无显著性意义(P > 0.05)。而急性肺损伤组和急性肺损伤+CH4组中棕黄色凋亡细胞核出现率明显高于空白组(P < 0.05),而急性肺损伤+CH4组与急性肺损伤组相比,凋亡细胞的比例明显降低(P < 0.05)。 肺组织Western blot检测caspase-3蛋白在空白组与对照组中的表达无明显差异(P > 0.05),见图8,这与TUNEL结果相符。而在急性肺损伤组和急性肺损伤+CH4组中其表达明显增加(P < 0.05)。急性肺损伤+CH4组与急性肺损伤组相比,caspase-3的表达量显著降低(P < 0.05)。由以上结果可知,甲烷对正常大鼠肺组织的细胞凋亡没有显著影响,急性肺损伤造模可以显著的增加肺组织细胞的凋亡,饱和甲烷生理盐水处理可以显著的降低肺组织细胞的凋亡。"
| [1] Ladha K, Vidal Melo MF, McLean DJ, et al.Intraoperative protective mechanical ventilation and risk of postoperative respiratory complications: hospital based registry study.BMJ. 2015;351:h3646.[2] Gu WJ, Wan YD, Tie HT, et al.Risk of acute lung injury/acute respiratory distress syndrome in critically ill adult patients with pre-existing diabetes: a meta-analysis.PLoS One. 2014;9(2): e90426.[3] Londhe C, Ganeriwal A, deSouza R.Study of clinical profile of acute respiratory distress syndrome and acute lung injury in Plasmodium vivax malaria.J Vector Borne Dis. 2014;51(4): 339-342.[4] Luhr OR, Antonsen K, Karlsson M,et al.Incidence and mortality after acute respiratory failure and acute respiratory distress syndrome in Sweden, Denmark, and Iceland. The ARF Study GroupAm J Respir Crit Care Med. 1999;159(6):1849-1861.[5] Irish Critical Care Trials Group.Acute lung injury and the acute respiratory distress syndrome in Ireland: a prospective audit of epidemiology and management.Crit Care. 2008;12(1):R30. .[6] Lewandowski K, Metz J, Deutschmann C,et al.Incidence, severity, and mortality of acute respiratory failure in Berlin, Germany.Am J Respir Crit Care Med. 1995;151(4):1121-1125.[7] Johnson ER, Matthay MA.Acute lung injury: epidemiology, pathogenesis, and treatment.J Aerosol Med Pulm Drug Deliv. 2010;23(4):243-252.[8] Sawa T.The molecular mechanism of acute lung injury caused by Pseudomonas aeruginosa: from bacterial pathogenesis to host response.J Intensive Care. 2014;2(1):10.[9] Xie K, Yu Y, Pei Y,et al.Protective effects of hydrogen gas on murine polymicrobial sepsis via reducing oxidative stress and HMGB1 release.Shock. 2010;34(1):90-97.[10] Kohama K, Yamashita H, Aoyama-Ishikawa M, et al.Hydrogen inhalation protects against acute lung injury induced by hemorrhagic shock and resuscitation.Surgery. 2015;158(2): 399-407.[11] Zhang Y, Liu Y, Zhang J.Saturated hydrogen saline attenuates endotoxin-induced lung dysfunction.J Surg Res. 2015;198(1): 41-49.[12] Cloarec D, Bornet F, Gouilloud S, et al.Breath hydrogen response to lactulose in healthy subjects: relationship to methane producing status.Gut. 1990;31(3):300-304.[13] Jo JY, Kwon YS, Lee JW, et al.Acute respiratory distress due to methane inhalation.Tuberc Respir Dis (Seoul). 2013;74(3): 120-123.[14] Liu W, Wang D, Tao H, et al.Is methane a new therapeutic gas? Med Gas Res. 2012;2(1):25. .[15] Ye Z, Chen O, Zhang R,et al.Methane Attenuates Hepatic Ischemia/Reperfusion Injury in Rats Through Antiapoptotic, Anti-Inflammatory, and Antioxidative Actions.Shock. 2015; 44(2): 181-187.[16] Matute-Bello G, Frevert CW, Martin TR.Animal models of acute lung injury.Am J Physiol Lung Cell Mol Physiol. 2008;295(3): L379-99.[17] Choi CW, Lee J, Oh JY, et al.Protective effect of chorioamnionitis on the development of bronchopulmonary dysplasia triggered by postnatal systemic inflammation in neonatal rats.Pediatr Res. 2016;79(2):287-294.[18] Lee HJ, Lee YJ, Jo HS,et al.Double exposure to intra-amniotic lipopolysaccharide and maternal betamethasone induces sustained increase of neutrophils in the lungs and disrupts alveolarization in newborn rats.J Perinat Med. 2013;41(6): 711-718.[19] Rubenfeld GD, Caldwell E, Peabody E,et al.Incidence and outcomes of acute lung injury.N Engl J Med. 2005;353(16): 1685-1693.[20] Huang Y, Pettitt SJ, Guo G, et al. Isolation of homozygous mutant mouse embryonic stem cells using a dual selection system. Nucleic Acids Res. 2012;40(3):e21.[21] Aulakh GK, Suri SS, Singh B.Angiostatin inhibits acute lung injury in a mouse model.Am J Physiol Lung Cell Mol Physiol. 2014; 306(1):L58-68.[22] Xie K, Yu Y, Huang Y, et al.Molecular hydrogen ameliorates lipopolysaccharide-induced acute lung injury in mice through reducing inflammation and apoptosis.Shock. 2012;37(5):548-555.[23] Liu H, Yu X, Yu S, et al.Molecular mechanisms in lipopolysaccharide-induced pulmonary endothelial barrier dysfunction.Int Immunopharmacol. 2015;29(2):937-946.[24] Ward PA, Grailer JJAcute lung injury and the role of histones. Transl Respir Med. 2014;2:1.[25] Bhatia M, Moochhala S.Role of inflammatory mediators in the pathophysiology of acute respiratory distress syndrome.J Pathol. 2004;202(2):145-156.[26] Biswal S, Remick DG.Sepsis: redox mechanisms and therapeutic opportunities.Antioxid Redox Signal. 2007;9(11):1959-1961 .[27] Qi XF, Teng YC, Yoon YS,et al.Reactive oxygen species are involved in the IFN-gamma-stimulated production of Th2 chemokines in HaCaT keratinocytes.J Cell Physiol. 2011;226(1): 58-65.[28] Tajima S, Soda M, Bando M, et al.Preventive effects of edaravone, a free radical scavenger, on lipopolysaccharide-induced lung injury in mice.Respirology. 2008;13(5):646-653.[29] Gwarzo MY, Muhammad SA.Extracellular superoxide dismutase activity and plasma malondialdehyde in human immunodeficiency virus subjects of kano state as surrogate markers of CD4 status. Int J Biomed Sci. 2010;6(4):294-300.[30] Hassett P, Curley GF, Contreras M, et al.Overexpression of pulmonary extracellular superoxide dismutase attenuates endotoxin-induced acute lung injury.Intensive Care Med. 2011; 37(10):1680-1687.[31] Song Y, Mao B, Qian G.Effects of dexamethasone on apoptosis and expression of Fas/FasL system in lung tissues of ALI rats.Zhonghua Jie He He Hu Xi Za Zhi. 2000;23(1):23-26.[32] Nakamura M, Matute-Bello G, Liles WC,et al.Differential response of human lung epithelial cells to fas-induced apoptosis.Am J Pathol. 2004;164(6):1949-1958.[33] Chen X, Liu Q, Wang D,et al.Protective Effects of Hydrogen-Rich Saline on Rats with Smoke Inhalation Injury.Oxid Med Cell Longev. 2015;2015:106836.[34] Zhao LL, Hu GC, Zhu SS, et al.Propofol pretreatment attenuates lipopolysaccharide-induced acute lung injury in rats by activating the phosphoinositide-3-kinase/Akt pathway.Braz J Med Biol Res. 2014 Oct 14;0:0. [Epub ahead of print].[35] Song K, Zhang M, Hu J,et al.Methane-rich saline attenuates ischemia/reperfusion injury of abdominal skin flaps in rats via regulating apoptosis level.BMC Surg. 2015;15:92.[36] Wu J, Wang R, Ye Z, et al.Protective effects of methane-rich saline on diabetic retinopathy via anti-inflammation in a streptozotocin-induced diabetic rat model.Biochem Biophys Res Commun. 2015;466(2):155-161.[37] Pimentel M, Lin HC, Enayati P, et al.Methane, a gas produced by enteric bacteria, slows intestinal transit and augments small intestinal contractile activity.Am J Physiol Gastrointest Liver Physiol. 2006 Jun;290(6):G1089-1095.[38] Wang W, Huang X, Li J, et al.Methane Suppresses Microglial Activation Related to Oxidative, Inflammatory, and Apoptotic Injury during Spinal Cord Injury in Rats.Oxid Med Cell Longev. 2017;2017:2190897. [39] Boros M, Ghyczy M, Érces D,et al.The anti-inflammatory effects of methane.Crit Care Med. 2012;40(4):1269-1278.[40] Kai T, Jones KA, Warner DO.Halothane attenuates calcium sensitization in airway smooth muscle by inhibiting G-proteins. Anesthesiology. 1998;89(6):1543-1552.[41] Fink MP.Pharmacological effects of inhaled methane: plausible or not?.Crit Care Med.2012;4:1379-1380.中国组织工程研究杂志出版内容重点:组织构建;骨细胞;软骨细胞;细胞培养;成纤维细胞;血管内皮细胞;骨质疏松;组织工程 |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Li Jun, Zuo Xinhui, Liu Xiaoyuan, Zhang Kai, Han Xiangzhen, He Huiyu, . Effect of over expression of miR-378a on osteogenic and vascular differentiation of bone marrow mesenchymal stem cell sheet [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(31): 4939-4944. |
| [11] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||